
by codm | Dec 18, 2023 | Newsletter
For Print (PDF)
Eisai Co., Ltd. (Headquarters: Tokyo, CEO: Haruo Naito, “Eisai”) announced today that it has submitted a marketing authorization application in Japan for its in-house discovered fibroblast growth factor (FGF) receptor (FGFR1, FGFR2, FGFR3) selective tyrosine kinase inhibitor tasurgratinib succinate (generic name, development code: E7090, “tasurgratinib”) for biliary tract cancer with FGFR2 gene fusion. In Japan, tasurgratinib has received orphan drug designation for a prospective indication for unresectable biliary tract cancer with FGFR2 gene fusion by the Ministry of Health, Labour and Welfare, (MHLW). Under this system, this application will be subject to priority review.
This application is based on the results of a multicenter, open-label, single-arm clinical phase II trial (Study 201) in Japan and China conducted by Eisai. Study 201 enrolled patients with unresectable biliary tract cancer with FGFR2 gene fusion previously treated with gemcitabine-based combination chemotherapy. The primary endpoint of this study was objective response rate, and secondary endpoints included safety. Detailed results of the study will be presented at upcoming academic conferences.
The estimated number of patients with biliary tract cancer is approximately 25,000 in Japan1, 2 and the five-year survival rate for the cancer is approximately 25%, which makes it an intractable cancer with the second worst prognosis following pancreatic cancer.1 Drug therapy options are limited in comparison with other cancers, and as such it is a disease with significant unmet medical needs. FGFR2 gene fusion is observed in approximately 14% of intrahepatic cholangiocarcinoma, which account for 15-30% of biliary tract cancers.3 FGFR genetic aberrations such as the gene fusions are known to be deeply involved in the proliferation, survival and migration of cancer cells as well as tumor angiogenesis and drug resistance. As these genetic aberrations in FGFRs have been observed in various other types of cancers including biliary tract cancer, there is growing interest in FGFRs as a promising target for cancer therapy. By selectively inhibiting FGFR1, 2 and 3, and blocking those signals, tasurgratinib has been expected to become a new molecular targeted therapy for cancers with FGFR genetic aberrations.4
Eisai acknowledges “Oncology” as one of its key strategic areas, and will continue to focus on the discovery and development of anti-cancer drugs within drug discovery domains including “tumor microenvironment”, “proteostasis disruption”, “cell linage and cell differentiation”, and “inflammation, hypoxia, oxidative stress and cell senescence” under the Deep Human Biology Learning (DHBL) drug discovery and development organization. Eisai aspires to discover innovative new drugs with new targets and mechanisms of action from these domains with the aim of contributing to the cure of cancers.
Media Inquiries:
Public Relations Department,
Eisai Co., Ltd.
+81-(0)3-3817-5120
[Notes to editors]
1. About tasurgratinib (development code: E7090)
Discovered in-house by Eisai’s Tsukuba Research Laboratories, tasurgratinib is an orally available novel tyrosine kinase inhibitor that demonstrates selective inhibitory activity against fibroblast growth factor receptors (FGFR) FGFR1, FGFR2 and FGFR3. Distinct from prior known FGFR inhibitors, tasurgratinib has a basic structure which lacks the dimethoxyphenyl moiety, and in a kinetic interaction analysis study, it was observed that tasurgratinib demonstrates antitumor effects due to inhibition of kinase activity with a binding mode (Type V) that exhibits rapid and potent binding as well as high selectivity to FGFR.4
In addition to a Phase II clinical trial (Study 201) of tasurgratinib in Japan and China to evaluate efficacy and safety in patients with cholangiocarcinoma with FGFR2 gene fusion, a Phase I clinical trial is underway in Japan in patients with estrogen receptor-positive and HER2-negative breast cancer.
1.Latest statistics, Cancer Information Service, National Cancer Center, Japan.
2.The 23rd Follow-up Survey Reports for Primary Liver Cancer Cases in Japan (2014-2015), 2023.
3.Arai Y. et al., “Fibroblast growth factor receptor 2 tyrosine kinase fusions define a unique molecular subtype of cholangiocarcinoma”, Hepatology, 2014, 59, 1427-1434.
4.Miyano SW. et al., “E7090, a Novel Selective Inhibitor of Fibroblast Growth Factor Receptors, Displays Potent Antitumor Activity and Prolongs Survival in Preclinical Models”, Molecular Cancer Therapeutics, 2016, 15, 2630-2639.

by codm | Dec 13, 2023 | Newsletter
For Print (PDF)
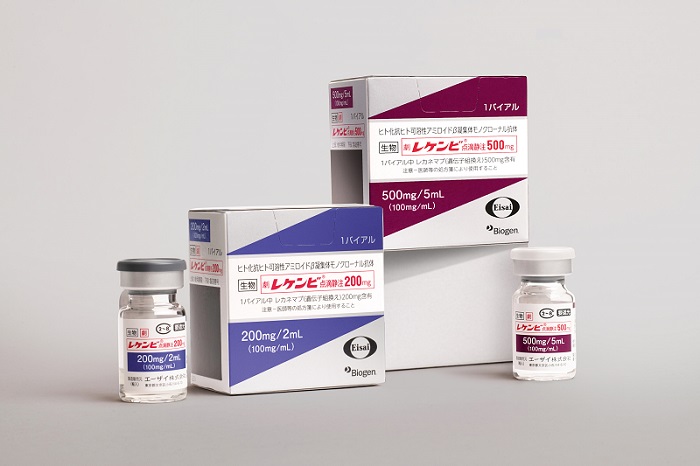
TOKYO and CAMBRIDGE, Mass., December 13, 2023 – Eisai Co., Ltd. (Headquarters: Tokyo, CEO: Haruo Naito, “Eisai”) and Biogen Inc. (Nasdaq: BIIB, Corporate headquarters: Cambridge, Massachusetts, CEO: Christopher A. Viehbacher, “Biogen”) announced today that humanized anti- soluble aggregated amyloid-beta (Aβ) monoclonal antibody “LEQEMBI® Intravenous Infusion” (200 mg, 500mg, lecanemab) will be launched in Japan on December 20, following its scheduled inclusion in the price listing on the Japan National Health Insurance (NHI) Drug Price List.
LEQEMBI received manufacturing and marketing approval for the indication of slowing progression of mild cognitive impairment (MCI) and mild dementia due to Alzheimer’s disease (AD) in Japan on September 25, 2023. In addition to inclusion in Japan’s NHI Drug Price List, the product’s Optimal Clinical Use Guidelines were agreed at a general meeting of the Central Social Insurance Medical Council, an advisory body of the Japanese Ministry of Heath, Labour and Welfare, held today. The launch in Japan marks the second country to have LEQEMBI on the market, following the U.S.
In Japan, Eisai and Biogen Japan will co-promote LEQEMBI, with Eisai distributing the product as the Marketing Authorization Holder.
LEQEMBI selectively binds to soluble amyloid-beta (Aβ) aggregates (protofibrils), as well as insoluble Aβ aggregates (fibrils) which are a major component of Aβ plaques, thereby reducing both Aβ protofibrils and Aβ plaques in the brain. LEQEMBI is the first and only approved treatment shown to reduce the rate of disease progression and to slow cognitive and functional decline through this mechanism.
“I am keenly aware of the weight of our responsibility moving forward as we launch LEQEMBI, the world first anti-amyloid Alzheimer’s disease treatment shown to slow the progress of the disease, in Japan, where Eisai’s value creation has started” said Haruo Naito, Chief Executive Officer at Eisai. “The establishment of an optimal and fast Alzheimer’s disease diagnosis and treatment pathway for patients is a top priority, and close collaboration among the government, dementia specialists, primary care physicians, radiologists, pharmacists, nurses, clinical psychologists, radiology staff, medical office personnel, and caregivers is essential for this purpose. In consideration of the importance of Alzheimer’s disease in Japan, we believe it is imperative that such pathways be established. We are committed to taking this first step towards changing the future of Alzheimer’s disease together with our stakeholders.”
“The availability of LEQEMBI opens a new era in the treatment of Alzheimer’s disease potentially giving patients and their families additional valuable time together and further positions Japan as a leader in caring for an elderly population,” said Christopher A. Viehbacher, President and Chief Executive Officer of Biogen “We will work alongside Eisai to engage the medical community and support the patient journey, especially early diagnosis, as mounting evidence suggests early intervention may provide greater impact on disease progression.”
Eisai will conduct a post-marketing special use results survey in all patients who are administered LEQEMBI (all-case surveillance) until data from a certain number of patients are accumulated, in accordance with an approval condition imposed by the Ministry of Health, Labour and Welfare. In addition, the appropriate use of LEQEMBI will be promoted in accordance with the package insert and the Optimal Clinical Use Guidelines, and training materials will be provided for healthcare professionals to assist with the management and monitoring of amyloid-related imaging abnormalities (ARIA).
Eisai and Biogen are committed to promoting the early detection and diagnosis of AD towards its early treatment, and will do their utmost to deliver LEQEMBI the people with early AD and realize a Dementia-Inclusive Society.
*Protofibrils are large Aβ aggregated soluble species of 75-5000 Kd.1,2,3
MEDIA CONTACTS
| Eisai |
Biogen |
|
Eisai Co., Ltd.
Public Relations Department
TEL: +81 (0)3-3817-5120
Eisai Inc. (U.S.)
Libby Holman
+ 1-201-753-1945
Libby_Holman@eisai.com
Eisai Europe, Ltd.
(UK, Europe, Australia, New Zealand and Russia)
EMEA Communications Department
+44 (0) 786 601 1272
EMEA-comms@eisai.net
TEL:03-3817-5120
FAX:03-3811-3077
|
|
INVESTOR CONTACTS
| Eisai |
Biogen |
|
Eisai Co., Ltd.
Investor Relations Department
TEL: +81 (0) 3-3817-5122
|
|
Notes to Editors
1. Product Outline in Japan
Product name: LEQEMBI® Intravenous Infusion 200mg, LEQEMBI® Intravenous Infusion 500mg
Generic name: Lecanemab (recombinant)
Indication for use: Slowing progression of mild cognitive impairment (MCI) and mild dementia due to Alzheimer’s disease.
Dosage and administration: The usual dose of lecanemab (recombinant) is 10mg/kg infused intravenously over approximately 1 hour, once every 2 weeks.
National Health Insurance (NHI) Drug Price (Scheduled to be listed on December 20):
LEQEMBI Intravenous Infusion 200mg 45,777 JPY per vial
LEQEMBI Intravenous Infusion 500mg 114,443 JPY per vial
Packaging:
LEQEMBI Intravenous Infusion 200mg 2mL per vial
LEQEMBI Intravenous Infusion 500mg 5mL per vial
Warnings and Contraindications
1. Warning
1.1 Prior to initiating administration of this drug, sufficient information should be provided to patients and their families/caregivers about the occurrence rate of ARIA due to this drug, the risk of ARIA, tests necessary for risk management, and measures to be taken when ARIA occurs. This drug should be administered after being informed and obtaining their consent. Also, patients should be in-structed to immediately contact their attending physician if any abnormalities are observed.
1.2 Prior to initiating administration of this drug, sufficient information should be provided to patients and their families/caregivers about the occurrence rate of ARIA due to this drug, the risk of ARIA, tests necessary for risk management, and measures to be taken when ARIA occurs. This drug should be administered after being informed and obtaining their consent. Also, patients should be in-structed to immediately contact their attending physician if any abnormalities are observed.
2. Contraindications (This drug is contraindicated to the following patients.)
2.1 Patients with a history of serious hypersensitivity to the ingredients of this drug.
2.2 Patients with cerebral vasogenic edema confirmed be-fore the start of administration of this drug. [Due to the possible increased risk of ARIA]
2.3 Patients with 5 or more cerebral microhemorrhages, focal cerebral surface hemosiderosis or cerebral hemorrhage >1 cm in size confirmed before the start of administration of this drug. [Due to the possible increased risk of ARIA]
2. About LEQEMBI
LEQEMBI (lecanemab) is the result of a strategic research alliance between Eisai and BioArctic. LEQEMBI is a humanized immunoglobulin gamma 1 (IgG1) monoclonal antibody directed against aggregated soluble (protofibril) and insoluble forms of amyloid-beta (Aβ). LEQEMBI is an amyloid beta-directed antibody indicated as a disease-modifying treatment for Alzheimer’s disease (AD) in the U.S. The U.S. Food and Drug Administration (FDA) granted traditional approval on July 6, 2023. In the U.S., treatment with LEQEMBI should be initiated in patients with mild cognitive impairment or mild dementia stage of disease, the population in which treatment was initiated in clinical trials. There are no safety or effectiveness data on initiating treatment at earlier or later stages of the disease than were studied.
Please see full U.S. Prescribing Information for LEQEMBI, including Boxed WARNING.
In Japan, Eisai received approval from the Ministry of Health, Labour and Welfare (MHLW) on September 25, 2023, to manufacture and market lecanemab as a treatment for slowing progression of mild cognitive impairment (MCI) and mild dementia due to AD.
LEQEMBI’s approval was based on Phase 3 data from Eisai’s large, global Clarity AD clinical trial, in which LEQEMBI met its primary endpoint and all key secondary endpoints with statistically significant results and confirmed the clinical benefit of LEQEMBI. The primary endpoint was the global cognitive and functional scale, Clinical Dementia Rating Sum of Boxes (CDR-SB). In the Clarity AD clinical trial, treatment with LEQEMBI reduced clinical decline on CDR-SB by 27% at 18 months compared to placebo. In addition, the secondary endpoint from the AD Cooperative Study-Activities of Daily Living Scale for Mild Cognitive Impairment (ADCS MCI-ADL), which measures information provided by people caring for patients with AD, noted a statistically significant benefit of 37% compared to placebo. The ADCS MCI-ADL assesses the ability of patients to function independently, including being able to dress, feed themselves and participate in community activities. The most common adverse events (>10%) in the LEQEMBI group were infusion reactions, ARIA-H (combined cerebral microhemorrhages, cerebral macrohemorrhages, and superficial siderosis), ARIA-E (edema/effusion), headache, and fall. Full results of the Clarity AD study were presented at the Clinical Trials on Alzheimer’s Disease (CTAD) 2022 conference and simultaneously published in the peer-reviewed medical journal The New England Journal of Medicine(New Window) on November 29, 2022.
Eisai has also submitted applications for approval of lecanemab in 12 countries and regions, including EU and China. In China and Israel, the applications have been designated for priority review, and in Great Britain, lecanemab has been designated for the Innovative Licensing and Access Pathway (ILAP), which aims to reduce the time to market for innovative medicines.
Eisai has completed a lecanemab subcutaneous bioavailability study, and subcutaneous dosing of lecanemab is currently being evaluated in the Clarity AD (Study 301) open-label extension (OLE). A maintenance dosing regimen has been evaluated as part of Study 201.
Since July 2020 the Phase 3 clinical study (AHEAD 3-45) for individuals with preclinical AD, meaning they are clinically normal and have intermediate or elevated levels of amyloid in their brains, is ongoing. AHEAD 3-45 is conducted as a public-private partnership between the Alzheimer’s Clinical Trial Consortium that provides the infrastructure for academic clinical trials in AD and related dementias in the U.S, funded by the National Institute on Aging, part of the National Institutes of Health, Eisai and Biogen. Since January 2022, the Tau NexGen clinical study for Dominantly Inherited AD (DIAD), that is conducted by Dominantly Inherited Alzheimer Network Trials Unit (DIAN-TU), led by Washington University School of Medicine in St. Louis, is ongoing and includes lecanemab as the backbone anti-amyloid therapy.
3. About the Collaboration between Eisai and Biogen for AD
Eisai and Biogen have been collaborating on the joint development and commercialization of AD treatments since 2014. Eisai serves as the lead of LEQEMBI development and regulatory submissions globally with both companies co-commercializing and co-promoting the product and Eisai having final decision-making authority.
4. About the Collaboration between Eisai and BioArctic for AD
Since 2005, Eisai and BioArctic have had a long-term collaboration regarding the development and commercialization of AD treatments. Eisai obtained the global rights to study, develop, manufacture and market LEQEMBI for the treatment of AD pursuant to an agreement with BioArctic in December 2007. The development and commercialization agreement on the antibody LEQEMBI back-up was signed in May 2015.
5. About Eisai Co., Ltd.
Eisai’s Corporate Concept is “to give first thought to patients and people in the daily living domain, and to increase the benefits that health care provides.” Under this Concept (also known as human health care (hhc) Concept), we aim to effectively achieve social good in the form of relieving anxiety over health and reducing health disparities. With a global network of R&D facilities, manufacturing sites and marketing subsidiaries, we strive to create and deliver innovative products to target diseases with high unmet medical needs, with a particular focus in our strategic areas of Neurology and Oncology.
In addition, we demonstrate our commitment to the elimination of neglected tropical diseases (NTDs), which is a target (3.3) of the United Nations Sustainable Development Goals (SDGs), by working on various activities together with global partners.
For more information about Eisai, please visit www.eisai.com (for global headquarters: Eisai Co., Ltd.), and connect with us on X, LinkedIn and Facebook.
6. About Biogen
Founded in 1978, Biogen is a leading global biotechnology company that has pioneered multiple breakthrough innovations including a broad portfolio of medicines to treat multiple sclerosis, the first approved treatment for spinal muscular atrophy, two co-developed treatments to address a defining pathology of Alzheimer’s disease, the first treatment to target a genetic form of ALS, the first oral treatment approved for postpartum depression, and the first approved treatment for Friedreich’s ataxia. Biogen is advancing a pipeline of potential novel therapies across neurology, neuropsychiatry, specialized immunology and rare diseases and remains acutely focused on its purpose of serving humanity through science while advancing a healthier, more sustainable and equitable world.
The company routinely posts information that may be important to investors on its website at www.biogen.com. Follow Biogen on social media –Facebook, LinkedIn, X, YouTube.
Biogen Safe Harbor
This news release contains forward-looking statements, including statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, about the potential clinical effects of LEQEMBI; the potential benefits, safety and efficacy of LEQEMBI; potential regulatory discussions, submissions and approvals and the timing thereof; the treatment of Alzheimer’s disease; the anticipated benefits and potential of Biogen’s collaboration arrangements with Eisai; the potential of Biogen’s commercial business and pipeline programs, including LEQEMBI; and risks and uncertainties associated with drug development and commercialization. These statements may be identified by words such as “aim,” “anticipate,” “believe,” “could,” “estimate,” “expect,” “forecast,” “intend,” “may,” “plan,” “possible,” “potential,” “will,” “would” and other words and terms of similar meaning. Drug development and commercialization involve a high degree of risk, and only a small number of research and development programs result in commercialization of a product. Results in early-stage clinical studies may not be indicative of full results or results from later stage or larger scale clinical studies and do not ensure regulatory approval. You should not place undue reliance on these statements or the scientific data presented.
These statements involve risks and uncertainties that could cause actual results to differ materially from those reflected in such statements, including without limitation unexpected concerns that may arise from additional data, analysis or results obtained during clinical studies, including the Clarity AD clinical trial and AHEAD 3-45 study; the occurrence of adverse safety events; risks of unexpected costs or delays; the risk of other unexpected hurdles; regulatory submissions may take longer or be more difficult to complete than expected; regulatory authorities may require additional information or further studies, or may fail or refuse to approve or may delay approval of Biogen’s drug candidates, including LEQEMBI; actual timing and content of submissions to and decisions made by the regulatory authorities regarding LEQEMBI; uncertainty of success in the development and potential commercialization of LEQEMBI; failure to protect and enforce Biogen’s data, intellectual property and other proprietary rights and uncertainties relating to intellectual property claims and challenges; product liability claims; third party collaboration risks; and the direct and indirect impacts of the ongoing COVID-19 pandemic on Biogen’s business, results of operations and financial condition. The foregoing sets forth many, but not all, of the factors that could cause actual results to differ from Biogen’s expectations in any forward-looking statement. Investors should consider this cautionary statement as well as the risk factors identified in Biogen’s most recent annual or quarterly report and in other reports Biogen has filed with the U.S. Securities and Exchange Commission. These statements a speak only as of the date of this news release. Biogen does not undertake any obligation to publicly update any forward-looking statements.
References
1. https://www.alzforum.org/news/conference-coverage/lecanemab-sweeps-toxic-av-protofibrils-catches-eyes-trialists
2. Sehlin D, Englund H, Simu B, Karlsson M, Ingelsson M, Nikolajeff F, Lannfelt L, Pettersson FE. Large aggregates are the major soluble Aβ species in AD brain fractionated with density gradient ultracentrifugation. PLoS One. 2012;7(2):e32014. https://doi.org/10.1371/journal.pone.0032014 Epub 2012 Feb 15. PMID: 22355408; PMCID: PMC3280222.
3. Söderberg, L., Johannesson, M., Nygren, P. et al. Lecanemab, Aducanumab, and Gantenerumab — Binding Profiles to Different Forms of Amyloid-Beta Might Explain Efficacy and Side Effects in Clinical Trials for Alzheimer’s Disease. Neurotherapeutics. 2023;20:195-206. https://doi.org/10.1007/s13311-022-01308-6

by codm | Dec 13, 2023 | Newsletter
For Print (PDF)
Eisai Co., Ltd.
Mizuho Bank, Ltd.
Eisai Co., Ltd. (CEO: Haruo Naito, “Eisai”) and Mizuho Bank, Ltd. (President & CEO: Katsuhiko Kato, “Mizuho Bank”) announced today that the companies have signed a sustainability-linked syndicated loan agreement (“the agreement”), which comes into effect today.
Sustainability-linked loans are a type of loan which incentivizes borrowers to achieve sustainability performance targets (“SPTs”) by linking borrowing terms and conditions, such as interest rates, to the achievement of the SPTs, seeking to drive environmental and socially sustainable economic activities and economic growth.
Under the concept of human health care (hhc), Eisai aims to effectively achieve social good in the form of relieving anxiety over health and reducing health disparities. through activities such as innovative drug discovery in the fields of dementia, cancer, and global health. Particularly in the domain of dementia, Eisai has developed “LEQEMBI®”, a new drug for Alzheimer’s disease(AD), which obtained approval in the US and Japan this year. By delivering LEQEMBI to people with early AD around the world, Eisai aims not only to improve their health outcomes but also contribute to address social issues regarding AD, including long term nursing care.
Eisai has set social impact targets for LEQEMBI in the U.S. and Japan based on its social value in terms of its effects on health outcomes and on cost reductions in nursing care and other expenses, and has established these as the SPTs of this agreement. With the funds raised through this agreement, Eisai will promote the creation of social impact through the elimination of diseases of poverty (neglected tropical diseases, malaria, tuberculosis, etc.), in addition to the establishment of programs to offer LEQEMBI to low-income populations for free in the U.S. and emerging countries, and the building of dementia ecosystems.
This agreement has undergone a third-party review* by Rating and Investment Information, Inc. (R&I) regarding its compliance with the Sustainability-Linked Loan Principles issued by the Loan Market Association and other organizations, as well as the Green Loan and Sustainability-Linked Loan Guidelines issued by Japan’s Ministry of the Environment.
Mizuho Bank is committed to the Mizuho group’s purpose of “proactively innovate together with our clients for a prosperous and sustainable future”, with the desire to co-create a prosperous future with clients by supporting their endeavors and challenging ourselves. Mizuho’s new medium-term business plan, which began in FY2023, is a three-year journey connecting various initiatives and co-creating value-added solutions in order to support our clients and solve social issues. As part of this, we are seeking out ways to have positive social impacts in areas such as realization of a more inclusive society and improvement in Japan’s global competitiveness.
Mizuho Bank’s role in connecting initiatives and creating social impact and outcomes is to support sustainability transformation from multiple angles by working together to find solutions for the development of clients and society from a medium- to long-term perspective.
Mizuho Bank will continue working together with our clients to achieve sustainability transformation by providing sustainable finance that creates a flow of funds to address environmental and social issues, and by providing solutions that utilize knowledge and functions that go beyond finance.
Overview of Loan Terms
Borrower: Eisai
Method: Syndication
Arranger: Mizuho Bank
Lenders: 39 financial institutions
Loan amount: 50 billion yen
Agreement date: December 6, 2023
Drawdown date: December 13, 2023
* Please refer to the opinion provided by Rating and Investment Information, Inc. (R&I).
https://www.r-i.co.jp/en/rating/esg/sustainabilityfinance/index.html
Media Inquiries:
| Eisai Co., Ltd. |
Mizuho Bank, Ltd |
|
Public Relations Department
+81 (0)3-3817-5120
|
Public Relations Office
+81 (0)3-5252-6574
|

by codm | Dec 9, 2023 | Newsletter
For Print (PDF)
TOKYO and RAHWAY, NJ, Dec. 9, 2023 – Eisai (Headquarters: Tokyo, CEO: Haruo Naito) and Merck & Co., Inc., Rahway, NJ, USA (known as MSD outside of the United States and Canada) today announced that the Phase 3 LEAP-001 trial evaluating LENVIMA®, the orally available multiple receptor tyrosine kinase inhibitor discovered by Eisai, plus KEYTRUDA®, the anti-PD-1 therapy from Merck & Co., Inc., Rahway, NJ, USA, did not meet its dual primary endpoints of overall survival (OS) and progression-free survival (PFS) for the first-line treatment of patients with advanced or recurrent endometrial carcinoma whose disease is mismatch repair proficient (pMMR)/not microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) /MSI-H.
At the final analysis, KEYTRUDA plus LENVIMA did not improve OS or PFS sufficiently to meet the study’s prespecified statistical criteria in the first-line treatment of certain patients with advanced or recurrent endometrial carcinoma versus a standard of care, platinum-based chemotherapy doublet (carboplatin plus paclitaxel). The safety profile of LENVIMA plus KEYTRUDA was consistent with that observed in previously reported studies evaluating the combination. A full evaluation of the data from this study is ongoing. The companies will work with investigators to share the results with the scientific community.
“We remain confident in the proven benefit of KEYTRUDA plus LENVIMA for the treatment of appropriate patients with certain types of previously-treated advanced endometrial carcinoma based on results from the KEYNOTE-775/Study 309 trial and will continue to research the KEYTRUDA plus LENVIMA combination in patients with other types of difficult-to-treat cancers,” said Dr. Gregory Lubiniecki, Vice President, Global Clinical Development, Merck & Co., Inc., Rahway, NJ, USA Research Laboratories. “We are disappointed that the LEAP-001 trial did not reach its primary endpoints, as we had hoped to bring another potential treatment option to patients when first diagnosed with certain types of advanced or recurrent endometrial carcinoma.”
“Results from the LEAP-001 trial underscore the challenges of treating patients with advanced or recurrent endometrial carcinoma in the first-line setting,” said Dr. Corina Dutcus, Senior Vice President, Oncology Global Clinical Development Lead at Eisai Inc. “We remain optimistic about clinical development program for LENVIMA plus KEYTRUDA and are proud that the combination has become a standard of care option for patients with certain types of advanced or recurrent endometrial carcinoma whose disease has progressed following prior systemic therapy, and will continue our efforts to contribute to these patients. We are grateful to the patients, their loved ones, and the investigators whose participation is what makes scientific advancement possible.”
LENVIMA plus KEYTRUDA is approved in the U.S., the EU, Japan and other countries for the treatment of certain types of advanced endometrial carcinoma following prior systemic therapy in any setting and advanced renal cell carcinoma (RCC). Lenvatinib is marketed as KISPLYX® for advanced RCC in the EU. Eisai and Merck & Co., Inc., Rahway, NJ, USA are studying the LENVIMA plus KEYTRUDA combination through the LEAP (LEnvatinib And Pembrolizumab) clinical program in various tumor types, including but not limited to hepatocellular carcinoma, RCC, head and neck cancer, gastric cancer and esophageal cancer, across multiple clinical trials.
Results from the LEAP-001 trial do not affect the current approved indications for the KEYTRUDA plus LENVIMA combination or other ongoing trials from the LEAP clinical program.
Investor Contacts:
| Eisai Co., Ltd. |
Merck & Co., Inc., Rahway, NJ, USA |
|
Public Relations:
+81-(0)3-3817-5120
Investor Relations:
+81-(0) 3-3817-5122
|
Media Relations
Julie Cunningham: +1-(617) 519-6264
John Infanti: +1-(609) 500-4714
Investor Relations
Peter Dannenbaum: +1-(732) 594-1579
Damini Chokshi: +1-(732) 594-1577
|
About LEAP-001
LEAP-001 is a randomized, open-label Phase 3 trial (ClinicalTrials.gov, NCT03884101) evaluating LENVIMA plus KEYTRUDA versus carboplatin plus paclitaxel for the first-line treatment of advanced or recurrent endometrial carcinoma. The dual primary endpoints are PFS, as assessed by blinded independent central review (BICR) per Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST v1.1) modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ, and OS. The secondary endpoints include objective response rate, as assessed by BICR per RECIST v1.1, quality of life measures, and safety. The study enrolled an estimated 842 patients who were randomized 1:1 to receive:
・LENVIMA (20 mg orally once daily) plus KEYTRUDA (200 mg intravenously [IV] on Day 1 of each three-week cycle); or
・Paclitaxel (175 mg/m2 IV on Day 1 of each three-week cycle) plus carboplatin (IV infusion at a total dose of area-under-the-curve 6 [per Calvert’s formula] given on Day 1 of each three-week cycle).
About Endometrial Carcinoma
Endometrial carcinoma begins in the inner lining of the uterus, which is known as the endometrium and is the most common type of cancer in the uterus.1,2 Worldwide, it was estimated there were more than 417,000 new cases of uterine body cancer diagnosed and more than 97,000 deaths from the disease in 2020 (these estimates include both endometrial carcinomas and uterine sarcomas,3 more than 90% of uterine body cancers occur in the endometrium, so the actual numbers for endometrial carcinoma cases and deaths may be slightly lower than these estimates).4 In Japan, there were more than 17,000 new cases of uterine body cancer and more than 3,000 deaths from the disease in 2020.5 In the U.S., it is estimated there will be approximately 66,000 new cases of uterine body cancer diagnosed and approximately 13,000 deaths from the disease in 2023.6,7 In Europe, it is estimated there were more than 130,000 new cases of uterine body cancer and more than 29,000 deaths in 2020.3 The five-year relative survival rate for metastatic endometrial carcinoma (stage IV) is estimated to be approximately 20%.8
About LENVIMA® (lenvatinib) Capsules
LENVIMA, discovered and developed by Eisai, is an orally available multiple receptor tyrosine kinase inhibitor that inhibits the kinase activities of vascular endothelial growth factor (VEGF) receptors VEGFR1 (FLT1), VEGFR2 (KDR), and VEGFR3 (FLT4). LENVIMA inhibits other kinases that have been implicated in pathogenic angiogenesis, tumor growth, and cancer progression in addition to their normal cellular functions, including fibroblast growth factor (FGF) receptors FGFR1-4, the platelet derived growth factor receptor alpha (PDGFRα), KIT, and RET. In syngeneic mouse tumor models, LENVIMA decreased tumor-associated macrophages, increased activated cytotoxic T cells, and demonstrated greater antitumor activity in combination with an anti-PD-1 monoclonal antibody compared to either treatment alone. LENVIMA has been approved for the indications below.
Thyroid cancer
・Indication as monotherapy
(Approved in over 80 countries including Japan, the United States, China, and countries in Europe and Asia)
Japan: Unresectable thyroid cancer
The United States: The treatment of patients with locally recurrent or metastatic, progressive, radioiodine-refractory differentiated thyroid cancer (DTC)
Europe: The treatment of adult patients with progressive, locally advanced or metastatic, differentiated (papillary/follicular/Hürthle cell) thyroid carcinoma (DTC), refractory to radioactive iodine (RAI)
Hepatocellular carcinoma
・Indication as monotherapy
(Approved in over 80 countries including Japan, the United States, China, and countries in Europe and Asia)
Japan: Unresectable hepatocellular carcinoma
The United States: The first-line treatment of patients with unresectable hepatocellular carcinoma (HCC)
Europe: The treatment of adult patients with advanced or unresectable hepatocellular carcinoma (HCC) who have received no prior systemic therapy
Thymic carcinoma
・Indication as monotherapy (Approved in Japan)
Japan: Unresectable thymic carcinoma
Renal cell carcinoma (In Europe, the agent was launched under the brand name Kisplyx®)
・ Indication in combination with everolimus
(Approved in over 65 countries including the United States, and countries in Europe and Asia)
The United States: The treatment of adult patients with advanced renal cell carcinoma (RCC) following one prior anti-angiogenic therapy
Europe: The treatment of adult patients with advanced renal cell carcinoma following one prior vascular endothelial growth factor (VEGF) targeted therapy
・ Indication in combination with KEYTRUDA (generic name: pembrolizumab)
(Approved in over 45 countries including Japan, the United States, and countries in Europe and Asia)
Japan: Radically unresectable or metastatic renal cell carcinoma
The United States: The first-line treatment of adult patients with advanced renal cell carcinoma
Europe: The first-line treatment of adult patients with advanced renal cell carcinoma
Endometrial carcinoma
・Indication in combination with KEYTRUDA
(Approved [including conditional approval] in over 50 countries including Japan, the United States, and countries in Europe and Asia)
Japan: Unresectable, advanced or recurrent endometrial carcinoma that progressed after cancer chemotherapy
The United States: The treatment of patients with advanced endometrial carcinoma (EC) that is not microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation
Europe: The treatment of adult patients with advanced or recurrent endometrial carcinoma (EC) who have disease progression on or following prior treatment with a platinum-containing therapy in any setting and are not candidates for curative surgery
About KEYTRUDA® (pembrolizumab) Injection, 100mg
KEYTRUDA is an anti-programmed death receptor-1 (PD-1) therapy that works by increasing the ability of the body’s immune system to help detect and fight tumor cells. KEYTRUDA is a humanized monoclonal antibody that blocks the interaction between PD-1 and its ligands, PD-L1 and PD-L2, thereby activating T lymphocytes which may affect both tumor cells and healthy cells.
Merck & Co., Inc., Rahway, NJ, USA has the industry’s largest immuno-oncology clinical research program. There are currently more than 1,600 trials studying KEYTRUDA across a wide variety of cancers and treatment settings. The KEYTRUDA clinical program seeks to understand the role of KEYTRUDA across cancers and the factors that may predict a patient’s likelihood of benefitting from treatment with KEYTRUDA, including exploring several different biomarkers.
About the Eisai and Merck & Co., Inc., Rahway, NJ, USA Strategic Collaboration
In March 2018, Eisai and Merck & Co., Inc., Rahway, NJ, USA, known as MSD outside the United States and Canada, through an affiliate, entered into a strategic collaboration for the worldwide co-development and co-commercialization of LENVIMA. Under the agreement, the companies jointly develop, manufacture and commercialize LENVIMA, both as monotherapy and in combination with KEYTRUDA, the anti-PD-1 therapy from Merck & Co., Inc., Rahway, NJ, USA. Eisai and Merck & Co., Inc., Rahway, NJ, USA are studying the LENVIMA plus KEYTRUDA combination through the LEAP (LEnvatinib And Pembrolizumab) clinical program and are evaluating the combination in various tumor types across multiple clinical trials.
Eisai’s Focus on Cancer
Eisai acknowledges “Oncology” as one of its key strategic areas, and will continue to focus on the discovery and development of anti-cancer drugs within drug discovery domains including “microenvironment”, “proteostasis disruption”, “cell lineage and cell differentiation”, and “inflammation, hypoxia, oxidative stress and cell senescence” under the Deep Human Biology Learning (DHBL) drug discovery and development organization. Eisai aspires to discover innovative new drugs with new targets and mechanisms of action from these domains, with the aim of contributing to the cure of cancers.
About Eisai
Eisai’s Corporate Concept is “to give first thought to patients and people in the daily living domain, and to increase the benefits that health care provides.” Under this Concept [also known as our human health care (hhc) Concept], we aim to effectively achieve social good in the form of relieving anxiety over health and reducing health disparities. With a global network of R&D facilities, manufacturing sites and marketing subsidiaries, we strive to create and deliver innovative products to target diseases with high unmet medical needs, with a particular focus in our strategic areas of Neurology and Oncology.
In addition, our continued commitment to the elimination of neglected tropical diseases (NTDs), which is a target (3.3) of the United Nations Sustainable Development Goals (SDGs), is demonstrated by our work on various activities together with global partners.
For more information about Eisai, please visit www.eisai.com (for global headquarters: Eisai. Co., Ltd.), us.eisai.com (for U.S. headquarters: Eisai, Inc.) or www.eisai.eu (for Europe, Middle East, Africa, Russia, Australia and New Zealand headquarters: Eisai Europe Ltd.), and connect with us on X (U.S. and global), LinkedIn (for global, U.S. and EMEA) and Facebook (global).
Merck & Co., Inc., Rahway, NJ, USA’s Focus on Cancer
Our goal is to translate breakthrough science into innovative oncology medicines to help people with cancer worldwide. At Merck & Co., Inc., Rahway, NJ, USA, the potential to bring new hope to people with cancer drives our purpose and supporting accessibility to our cancer medicines is our commitment. As part of our focus on cancer, Merck & Co., Inc., Rahway, NJ, USA is committed to exploring the potential of immuno-oncology with one of the largest development programs in the industry across more than 30 tumor types. We also continue to strengthen our portfolio through strategic acquisitions and are prioritizing the development of several promising oncology candidates with the potential to improve the treatment of advanced cancers. For more information about our oncology clinical trials, visit www.merck.com/clinicaltrials.
About Merck & Co., Inc., Rahway, NJ, USA
For over 130 years, Merck & Co., Inc., Rahway, NJ, USA, known as MSD outside of the United States and Canada, has been inventing for life, bringing forward medicines and vaccines for many of the world’s most challenging diseases in pursuit of our mission to save and improve lives. We demonstrate our commitment to patients and population health by increasing access to health care through far-reaching policies, programs and partnerships. Today, Merck & Co., Inc., Rahway, NJ, USA continues to be at the forefront of research to prevent and treat diseases that threaten people and animals – including cancer, infectious diseases such as HIV and Ebola, and emerging animal diseases – as we aspire to be the premier research-intensive biopharmaceutical company in the world. For more information, visit www.merck.com and connect with us on Twitter, Facebook, Instagram, YouTube and LinkedIn.
Forward-Looking Statement of Merck & Co., Inc., Rahway, NJ, USA
This news release of Merck & Co., Inc., Rahway, NJ, USA (the “company”) includes “forward-looking statements” within the meaning of the safe harbor provisions of the U.S. Private Securities Litigation Reform Act of 1995. These statements are based upon the current beliefs and expectations of the company’s management and are subject to significant risks and uncertainties. There can be no guarantees with respect to pipeline candidates that the candidates will receive the necessary regulatory approvals or that they will prove to be commercially successful. If underlying assumptions prove inaccurate or risks or uncertainties materialize, actual results may differ materially from those set forth in the forward-looking statements.
Risks and uncertainties include but are not limited to, general industry conditions and competition; general economic factors, including interest rate and currency exchange rate fluctuations; the impact of the global outbreak of novel coronavirus disease (COVID-19); the impact of pharmaceutical industry regulation and health care legislation in the United States and internationally; global trends toward health care cost containment; technological advances, new products and patents attained by competitors; challenges inherent in new product development, including obtaining regulatory approval; the company’s ability to accurately predict future market conditions; manufacturing difficulties or delays; financial instability of international economies and sovereign risk; dependence on the effectiveness of the company’s patents and other protections for innovative products; and the exposure to litigation, including patent litigation, and/or regulatory actions.
The company undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events or otherwise. Additional factors that could cause results to differ materially from those described in the forward-looking statements can be found in the company’s Annual Report on Form 10-K for the year ended December 31, 2022 and the company’s other filings with the Securities and Exchange Commission (SEC) available at the SEC’s Internet site (www.sec.gov).
1 Mayo clinic, “Endometrial Cancer?”
https://www.mayoclinic.org/diseases-conditions/endometrial-cancer/symptoms-causes/syc-20352461#:~:text=Endometrial%20cancer%20begins%20in%20the%20layer%20of%20cells%20that%20form,less%20common%20than%20endometrial%20cancer.
2 American Cancer Society, “What Is Endometrial Cancer?”
https://www.cancer.org/cancer/endometrial-cancer/about/what-is-endometrial-cancer.html#:~:text=Endometrial%20cancer%20starts%20when%20cells,other%20parts%20of%20the%20body.
3 International Agency for Research on Cancer, World Health Organization. “Corpus uteri Fact Sheet.” Cancer Today, 2020.
https://gco.iarc.fr/today/data/factsheets/cancers/24-Corpus-uteri-fact-sheet.pdf
4 American Cancer Society, “Key Statistics for Endometrial Cancer.”
https://www.cancer.org/cancer/endometrial-cancer/about/key-statistics.html
5 International Agency for Research on Cancer, World Health Organization. “Japan Fact Sheet.” Cancer Today, 2020.
https://gco.iarc.fr/today/data/factsheets/populations/392-japan-fact-sheets.pdf
6 American Cancer Society, “Cancer Facts & Figures 2023.” https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2023/2023-cancer-facts-and-figures.pdf
7 Cancer Net ®.” Uterine Cancer”
https://www.cancer.net/cancer-types/uterine-cancer/view-all#:~:text=The%20average%20age%20of%20diagnosis,when%20it%20occurs%20after%20menopause.
8 American Cancer Society, “Survival Rates for Endometrial Cancer.”
https://www.cancer.org/cancer/endometrial-cancer/detection-diagnosis-staging/survival-rates.html

by codm | Nov 30, 2023 | Newsletter
For Print (PDF)
Eisai Co., Ltd. (Headquarters: Tokyo, CEO: Haruo Naito, “Eisai”) announced today that its Thai sales subsidiary Eisai (Thailand) Marketing Co., Ltd., (Eisai Thailand) has made an agreement to collaborate with the Department of Medical Services (DMS), Ministry of Public Health (MOPH) of Thailand to further enhance the access to treatments for dementia including Alzheimer’s disease (AD) in Thailand.
In Thailand, the number of people suffering from dementia is expected to increase significantly from the current 700 thousand to 2 million by 2050 due to the rapid aging of the population.1 Dementia and AD are the third leading cause of death among women (7.1% of total deaths) and the ninth leading cause of death for men (3.3% of total deaths),2 making the need to build a system to counter dementia urgent.
With this agreement, Eisai Thailand and DMS have identified three key challenges to addressing issues regarding dementia and AD in Thailand: (1) raising disease awareness for Mild Cognitive Impairment (MCI) and dementia including AD (2) improving diagnosis infrastructure for MCI and dementia including AD and ensuring civilian access to these services (3) ensuring total care by building a dementia community and ecosystem for people with dementia and their families.
Formulation of disease guidelines by the state is essential to address these challenges. Eisai Thailand and DMS will jointly conduct surveys and other initiatives to establish such guidelines. Additionally, both organizations will promote implementation of these guidelines in collaboration with various stakeholders, including those from different industries, to develop an ecosystem dedicated to dementia, seeking to establish a strong support system for the dementia community in Thailand.
Eisai will continue to support people with dementia and their families to “live their fullest lives” to effectively achieve social good in the form of “relieving anxiety over health” and “reducing health disparities”.
1 Alzheimer’s Disease International. & Alzheimer’s Australia. (2014). Dementia in the Asia Pacific Region. London: Alzheimer’s Disease International.
2 Burden of Disease Thailand Report 2019
Media Inquiries:
Public Relations Department,
Eisai Co., Ltd.
+81-(0)3-3817-5120







